Vinyl Ethers Radical
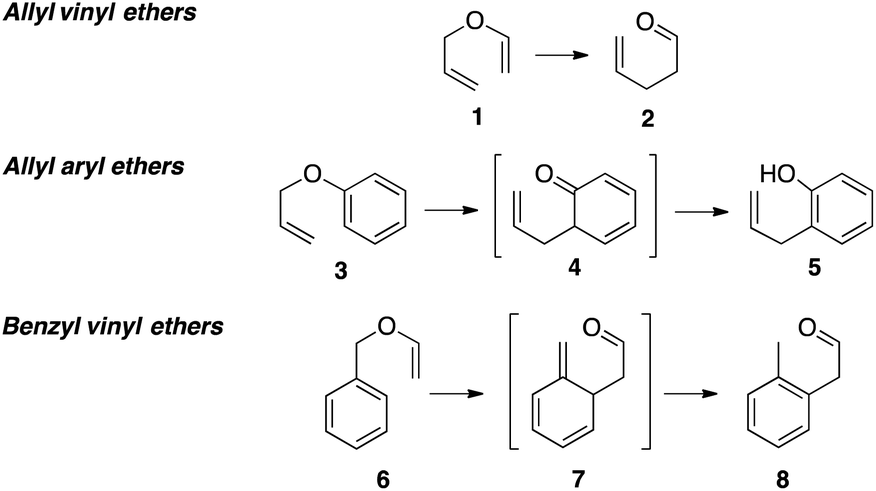
The radical produced by attack of a thiyl radical on the terminal methylene of the vinyl ether is stabilised by the oxygen atom of the ether group and therefore it is not surprising to find that vinyl ethers are very reactive ene components of the thiol ene reaction 4 the ether substituent on the double bond reduces the ionisation potential of.
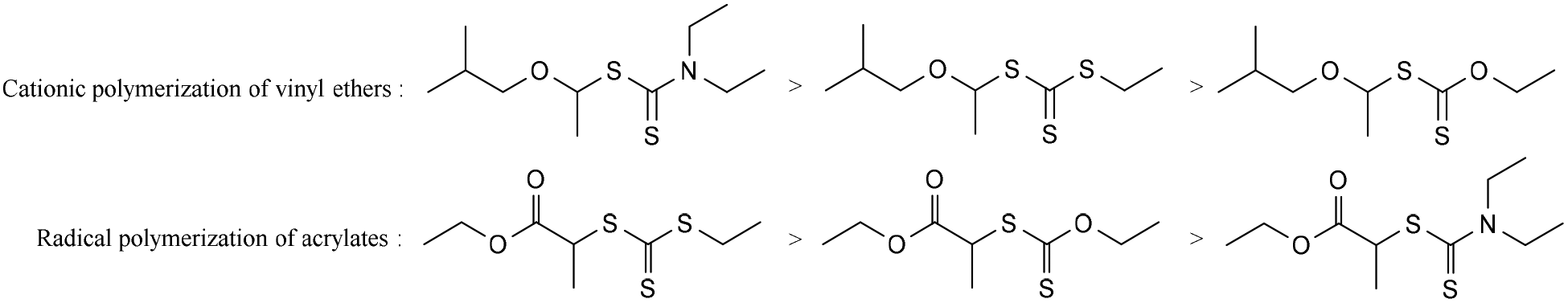
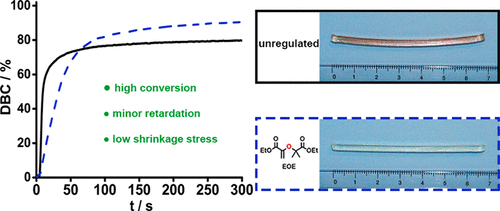
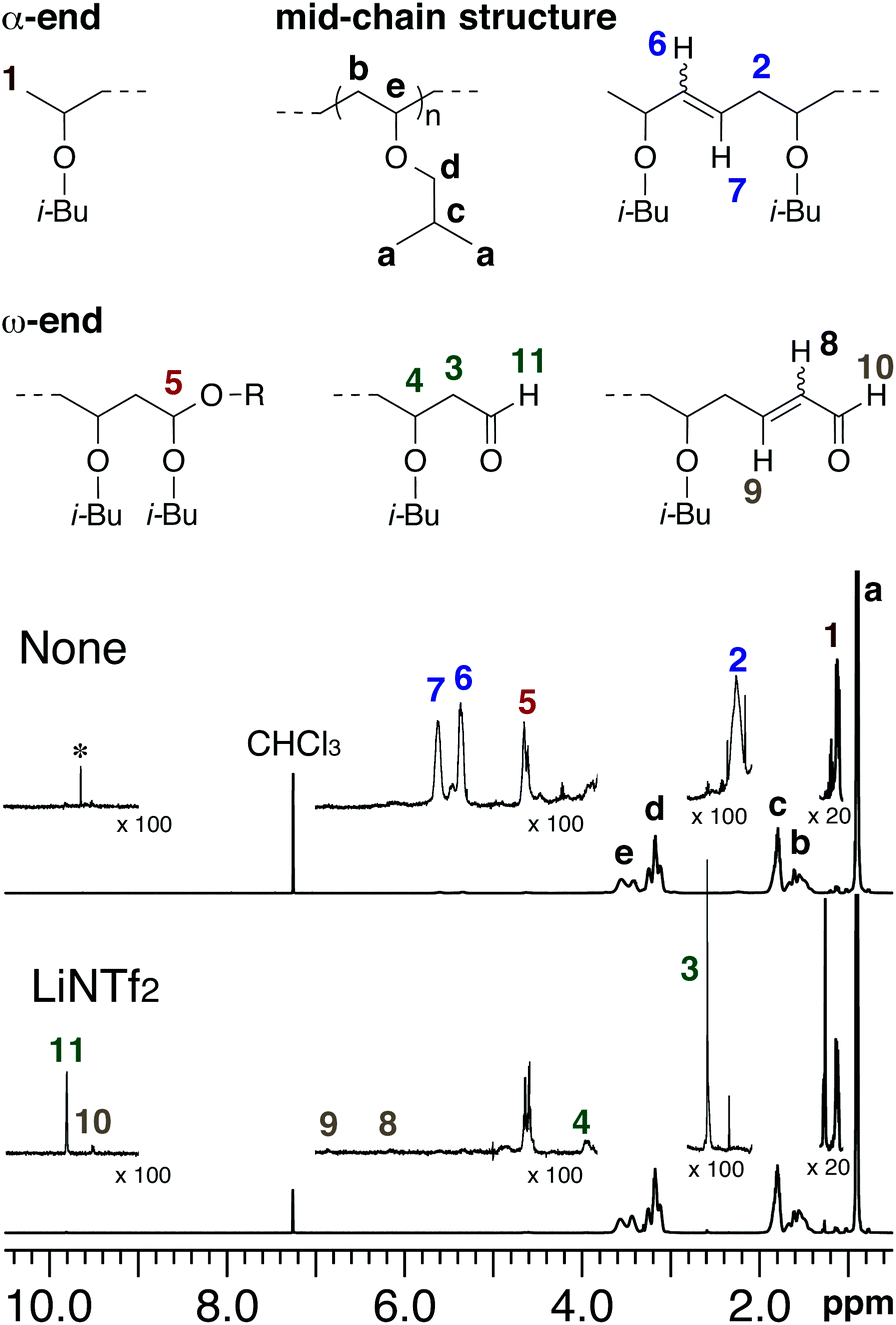
Vinyl ethers radical. Unprecedented controlled radical vinyl polymerization crp of vinyl ethers using reversible addition fragmentation chain transfer raft polymerization is reported. The product is used as low viscosity and fast curing monomers in cationic polymerization applications and as low viscosity monomers in radical polymerization applications. Teator and leibfarth report a general protocol to polymerize a variety of such vinyl ethers isotactically see the perspective by foster and o reilly. Polymerization rate in figure 5 the conversion of acrylate 1407 cm 1 and vinyl ether groups 1618 cm 1 are plotted as a function of vinyl ether content.
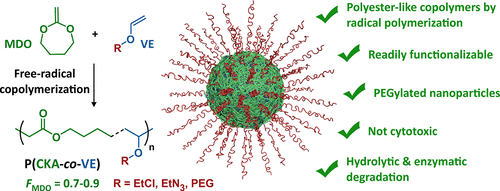
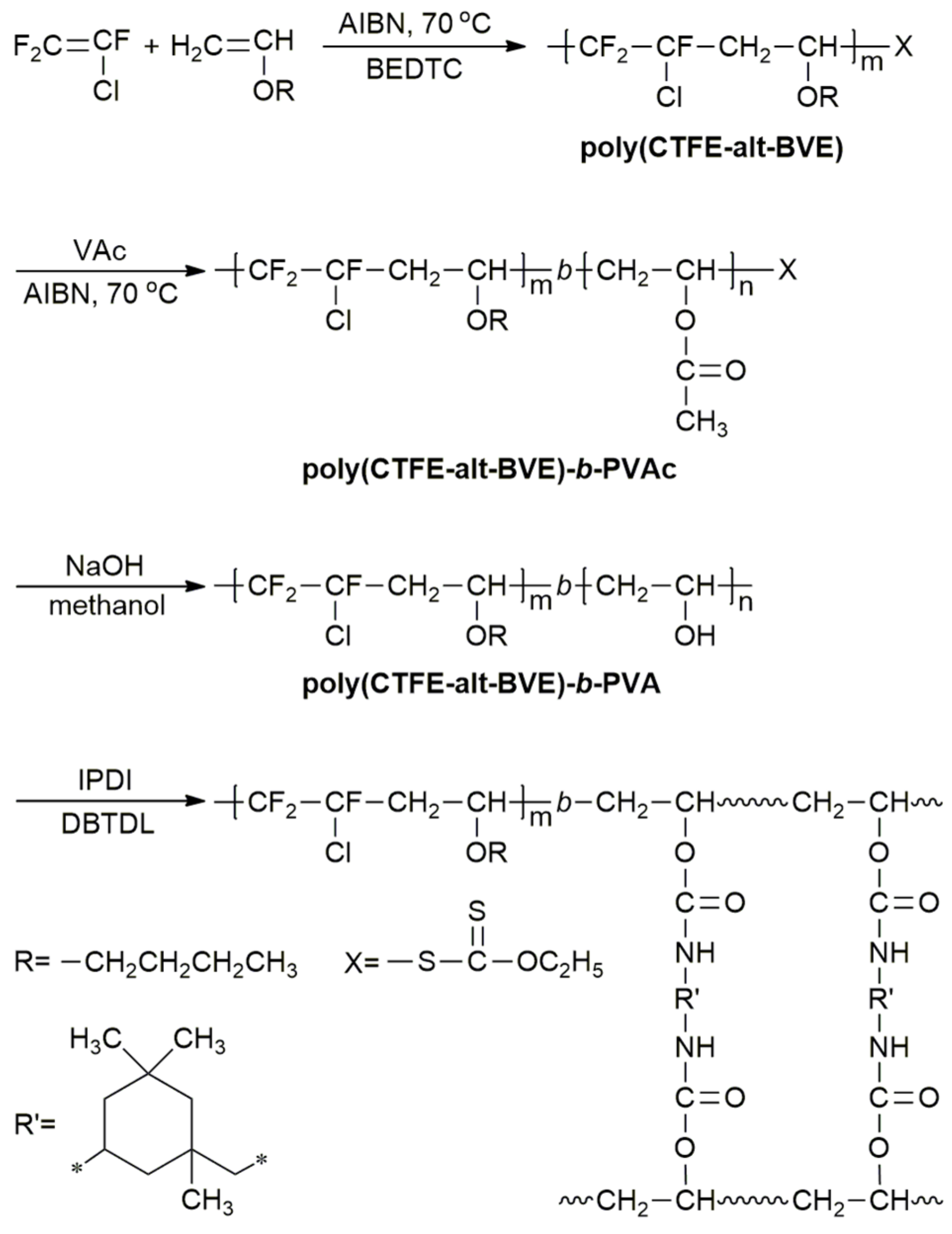
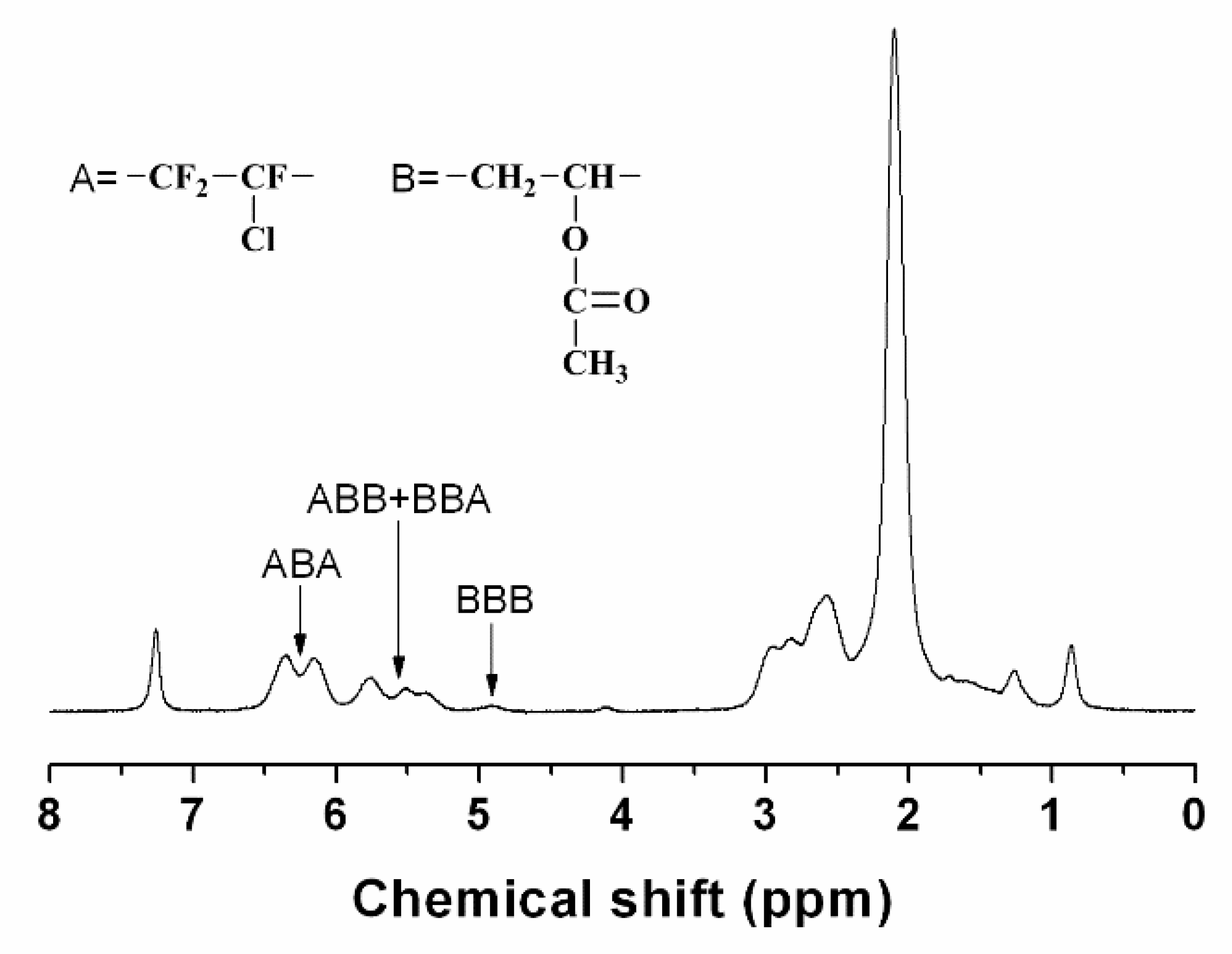
They are increasingly used in radiation curing systems because of a lower toxicity profile than the commonly used acrylic monomers. Free radical copolymerization of vinyl ethers is also possible. Add an oxygen into the monomer though and that degree of uniformity becomes harder to enforce. Copolymerization components include vinyl chloride vinyl acetate acrylic metacrylic acids and their ethers maleic anhydrides fluorinated olefin such as f2c cf2 and.
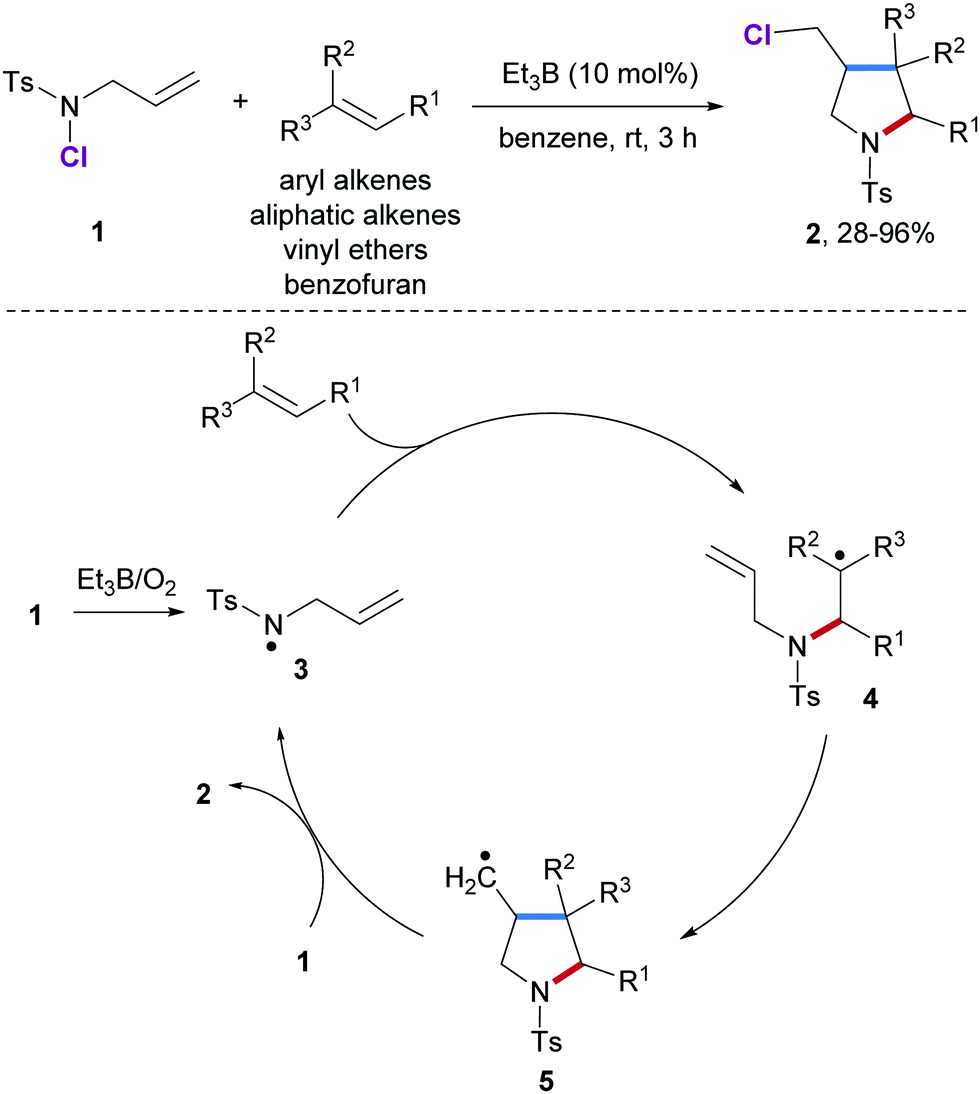
The thiol ene reaction also alkene hydrothiolation is an organic reaction between a thiol and an alkene to form a thioether this reaction was first reported in 1905 but it gained prominence in the late 1990s and early 2000s for its feasibility and wide range of applications. They rely on a chiral. Vinyl ether radical figure 5. It is remarkable that only 10 addition of dve 3 increases the conversion of the acrylate from.
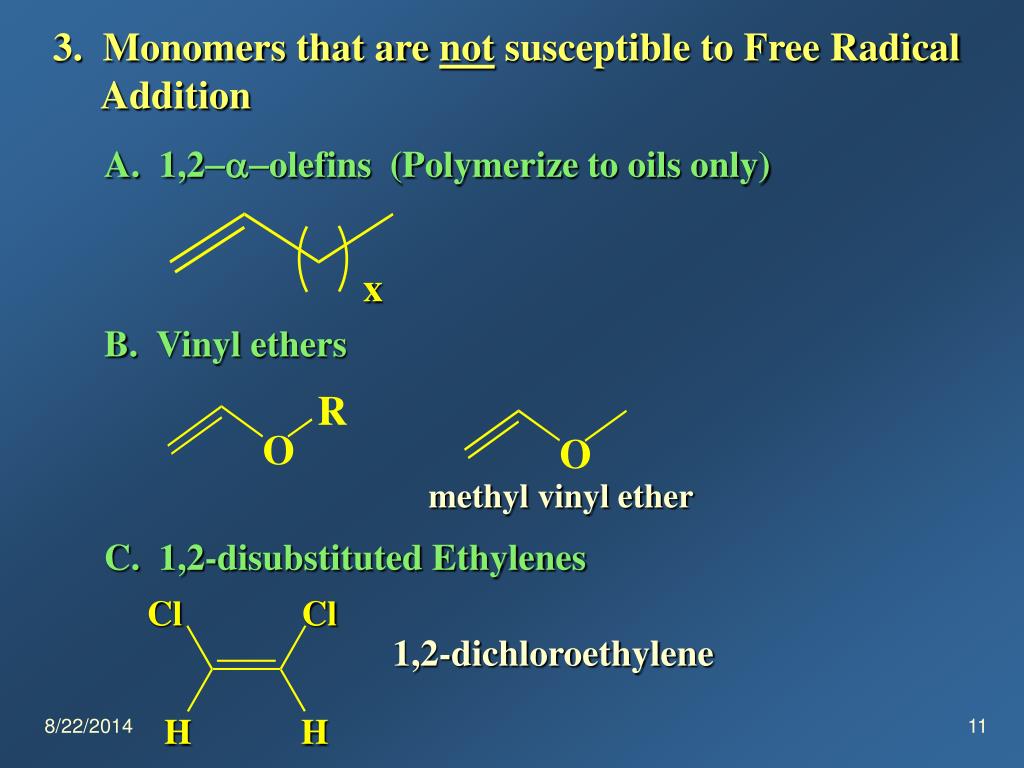
No anionic or radical polymerizations give high polymers from vinyl ethers except for the alternating radical copolymerization with an electron deficient monomer. Vinyl ethers 1 which have a strongly electron donating alkoxy substituent readily form polymers on treatment with an acidic compound initiator the polymerization is thus cationic in nature. Vinyl ethers undergo homopolymerization via a cationic mechanism. Furthermore in the presence of water they readily hydrolyse to acetaldehyde and.
However the polymerization under free radical conditions gives only low molecular weight oligomers and only copolymers can be synthesized. In order to overcome the challenge of direct radical polymerization of vinyl ethers commercial hydroxy functional vinyl ethers such as 2 hydroxyethyl vinyl ether heve were subjected to free radical polymerization generating. This reaction is accepted as a click chemistry reaction given the reactions high yield stereoselectivity high.